

CANaspire is an open-label clinical trial of an investigational gene therapy for Canavan disease. The trial is designed to assess the safety and potential benefit of the investigational gene therapy in Canavan patients.
Aspa’s investigational gene therapy trial uses an AAV9 vector (adeno-associated virus serotype 9), which is designed to deliver functional copies of the ASPA gene throughout the body, including into the brain, to address the underlying cause of Canavan disease.
The AAV9 vector Aspa is using for its investigational gene therapy has been studied in other rare disease gene therapy clinical trials, and is the vector used in the recently approved first gene therapy for spinal muscular atrophy, a pediatric neurodegenerative disease.
Evidence from human studies shows that the AAV9 vector when given intravenously (IV) gets into the central nervous system as well as tissues throughout the body.
Have a diagnosis and signs of Canavan disease
Be 30 months of age or younger on the expected date the investigational gene therapy would be given
Meet additional criteria (the study doctor will assess during screening)
There will be no cost to families for their child to receive the investigational gene therapy, including travel and other reasonable expenses associated with the clinical trial.
For more information about CANaspire please visit www.clinicaltrials.gov
Children with Canavan disease have mutations in the ASPA gene, which is responsible for making aspartoacylase, an enzyme that breaks down a chemical compound called N-acetyl-L-aspartic acid (NAA).
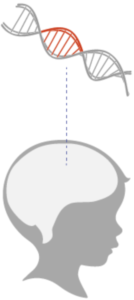
NAA is made mostly in neurons. If NAA is not broken down it accumulates in a child’s brain and may prevent the proper formation of myelin, the covering that protects neurons and helps neurons communicate with each other.
Deficits in myelin disrupt the health and function of neurons and are thought to cause Canavan’s effects on movement, language and vision.
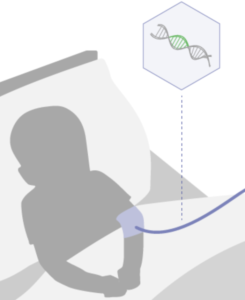
Aspa’s investigational gene therapy is given in a single intravenous dose using an AAV9 vector designed to deliver working copies of the ASPA gene throughout the body and the brain.
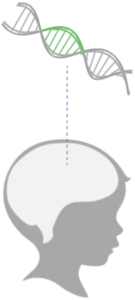
Once the functional ASPA gene is expressed, it may help restore myelin in the brain. This can potentially improve the course of Canavan disease by addressing the underlying genetic cause.
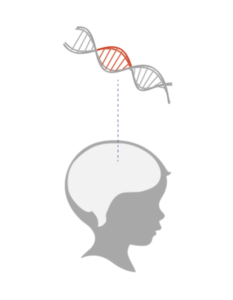
NAA is found mostly in neurons. If NAA is not broken down it accumulates in a child’s brain and may prevent the proper formation of myelin, the protective covering that helps neurons communicate with each other.
The deficits in myelin are thought to cause Canaan’s effects on movement, lanquage and vision.
Aspa’s investigational gene therapy is given in a single intravenous dose and aims to provide working copies of the
ASPA gene throughout a child’s body.
The treatment uses an AAV9 viral vector designed to deliver the functional ASPA gene throughout the body and the brain.
Once the functional ASPA gene is expressed, it may help restore myelin in the brain. This can potentially improve the course of Canavan disease by addressing the underlying genetic cause.
For more information about CANaspire please visit www.clinicaltrials.gov
To view a list of participating CANaspire sites and locations, please visit www.clinicaltrials.gov
Aspa’s gene therapy program is based on research by Guangping Gao, Ph.D. and Dominic J. Gessler, M.D., Ph.D. at the University of Massachusetts Medical School. Dr. Gao, a pioneer in AAV gene therapy, was also the first person to clone the ASPA gene in 1993, and has been working on developing a cure for Canavan disease for over 25 years.
Aspa is the first to initiate an international multi-center clinical trial using AAV9 gene therapy for children with Canavan disease.
You are leaving our website and we cannot be held responsible for the content of external websites.